 This isn't small change by any means, but the cost is offset to a great degree by the nicads' long life -- typically 5 to 7 times that of lead batteries (see graph). The table below details this. It's a cost analysis of two types of batteries that might be used for the Solectria Force. For comparison purposes, this chart also includes a cost analysis of a typical golf car battery. Golf car batteries are much too heavy for a Force, but many EV hobbyists use them in larger vehicles -- and as it turns out, golf car batteries are still the low-cost champs:
1. Optima Group 31 batteries are, for practical purposes, currently (mid-2001) unavailable. Nicads have advantages over lead batteries in most areas:
Environmental Considerations. Considerable concern has been expressed about the dangers of cadmium as a heavy metal, and much has been made of the disposal problem associated with nicad batteries. This is a significant issue for the millions of small flashlight cells used in rechargeable drills, vacuum cleaners, electric razors, and thousands of other appliances. Some responsibility for the problem can be laid on careless and/or poorly educated consumers. Probably more responsibility should be placed on the appliance manufacturers who design these gadgets to be cheap and disposable, encouraging the users to chuck them in the trash and buy new ones when their nicad batteries wear out. Usually, the only indication that users shouldn't do this is a fine-print warning on the appliance's carton, which has long since been discarded by the time the Dustbuster or other device has quit. The nicad cells in these small appliances cost a few dollars each. Not so with EV modules! While consumers may have little economic incentive to recycle their flashlight-size nicads, EV modules are worth considerably more. When the Saft modules finally wear out, it's cheaper to ship them back to Saft to be rebuilt than to buy new ones. And since Saft is required by European Union regulations to provide recycling services, they have the administrative structure in place to handle large-scale recycling. Anti-EV forces such as petroleum companies like to make much of the disposal and pollution issues associated with nicads (and with lead batteries), but the truth is that EV-scale batteries are an insignificant factor in these problems, and are likely to remain so.
Sources.
EV hobbyists can obtain
The Saft monoblocks in this project, however, weren't purchased new from Saft. They are used
modules, without warranty of course, obtained as surplus from an EV project in California. They
were originally put up in nicely-made
aluminum trays,
manufactured by Saft and fitted to
Pivco Citibees,
the forerunners to the
Charging.
Nickel-cadmium monoblocks require a somewhat different charging regimen than lead batteries.
In the first charging phase, they are charged at constant current until a temperature compensated
voltage threshold is reached or until all the amp-hours consumed in driving have been replaced,
whichever comes first. The charger notes how many amp-hours are added in this phase. Then,
in the second phase, a lower constant current finish charge is applied until a percentage of
the amp-hours in phase one has been added.
Obviously a smart charger is required. At this writing (late Spring 2001) one of the most significant
challenges to the hobbyist is finding a charger which implements the required algorithm. The
former easy answer was the
Maintenance.
As of now, all the nickel-cadmium modules of EV size are flooded batteries, so unlike valve
regulated lead batteries, they require some periodic maintenance.
Most flooded nicads, including earlier Saft types, are ferocious water users. The gassing voltage
level for nicads is quite low, so they always gas vigorously (disassociating the water of their electrolyte
into hydrogen and oxygen) during the charging finish stage.
However, the STM5-MR monoblocks not only need very infrequent watering -- a few times a year --
because, although they don't use an immobilized electrolyte, they do use a recombination catalyst
similar to that employed in valve-regulated lead
batteries. This catalyst, a bit of platinum in each cell, prompts the recombination of hydrogen and
oxygen back into water (evolving heat in the process).
For the occasions when they do need watering, a rather clever central
watering system makes the process relatively easy.
One watering circuit is required per 10 modules, since Saft stipulates that the potential from beginning
to end of the watering circuit shouldn't exceed 60 volts. Each circuit consists of modules connected
in "series," and the circuit thus has an inlet and an outlet.
When it's time to water, a special charge cycle is carried out to ensure that the electrolyte is at its
maximum point. There's no need to remove the cell caps (in fact there are none; the module tops
are sealed). A gravity-fed tank is attached to the water inlet on the first monoblock, and
distilled water is allowed to flow until the water runs from the outlet on the last monoblock. That's
all there is to it.
Besides the ease of maintenance, another advantage of this system is that it is fully closed. That
is, the watering system also provides hydrogen venting to the outside of the vehicle.Therefore,
there's no venting of gases or emission of electrolyte mist inside the battery boxes of the vehicle.
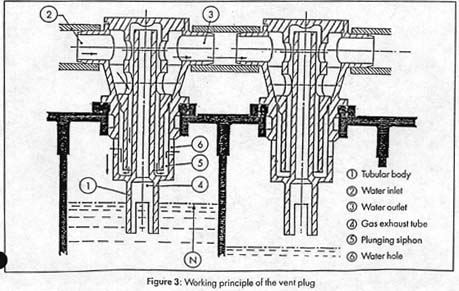
How it Works. The diagram below illustrates the principle. This shows a removeable vent
cap from an earlier Saft monoblock design, without the recombinant feature, but the principle for the
MR range of monoblocks is the same.
Here is a description of the system, adapted from the Saft technical manual:
The automatic vent caps have no moving parts and are essentially foolproof. The concept is to fill
a cell with water up to a specified level allowing gas which is in the cell to escape. When the
specified level is reached, the electrolyte closes the gas exhaust tube and the consequent excess
pressure stops the water flow into the cell. The water will now flow to the next cell and so on,
to the last cell of the hydraulic circuit.
Each vent cap has a tubular body (1) fitted with a water inlet (2) and outlet (3) in its upper part, and
with a gas exhaust tube (4) in its lower part. The water flows across the vent because of a plunging
siphon (5) and then flows into the cell through a water hole (6), while the gas escapes through the
exhaust tube (4).
The lower edge of the gas exhaust tube (4) determines the electrolyte level of the cell.
During the watering process, when the electrolyte reaches this level, the air under the cover can
no longer escape through the gas exhaust tube (4), and the water reserve generated by the
plunging siphon ensures a safe obstruction of the gas inside the cell. The water filling of the
cell is over, and the water flows to the next cell through the outlet (3).
In order for the system to work correctly, the water flow must be lower than 0.7 liter per minute and
the relative internal pressure below 0.15 bars.
|